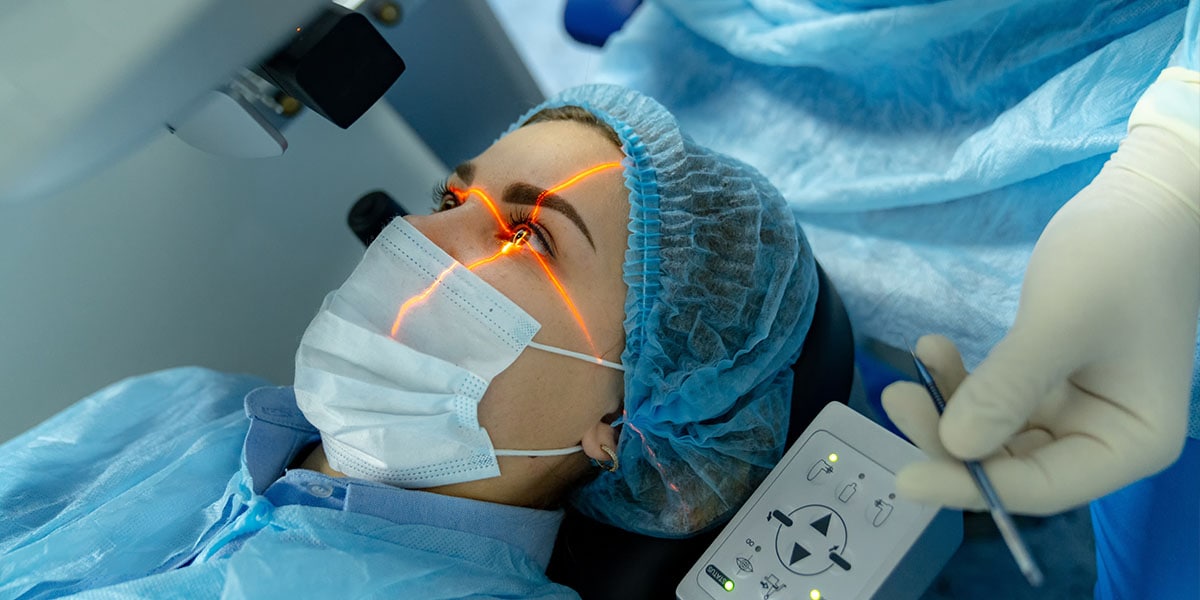
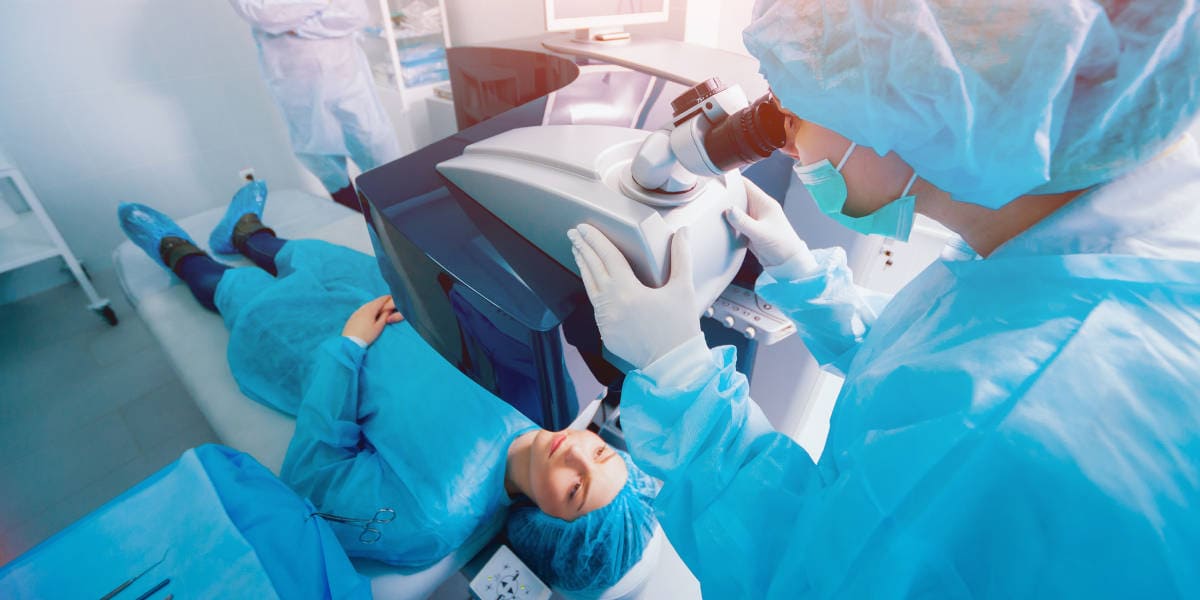
No two eyes are the same, so request a complimentary LASIK evaluation from our experts.
The latest advance in LASIK greatly enhances outcomes and reduces side effects.
The nationally recognized Center of Excellence for cataract care.
The most effective treatments from LA's award winning glaucoma surgeons.
Los Angeles's leading Vitreous & Retina Treatment Center.
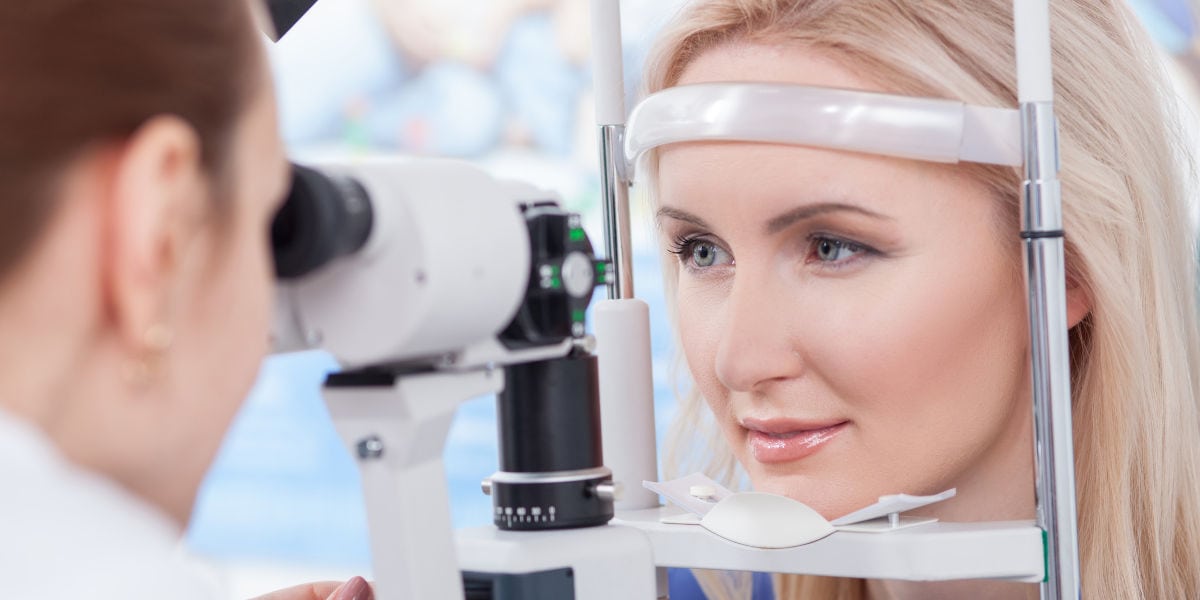
Trust Your Eyes to the Best Ophthalmologists in Los Angeles
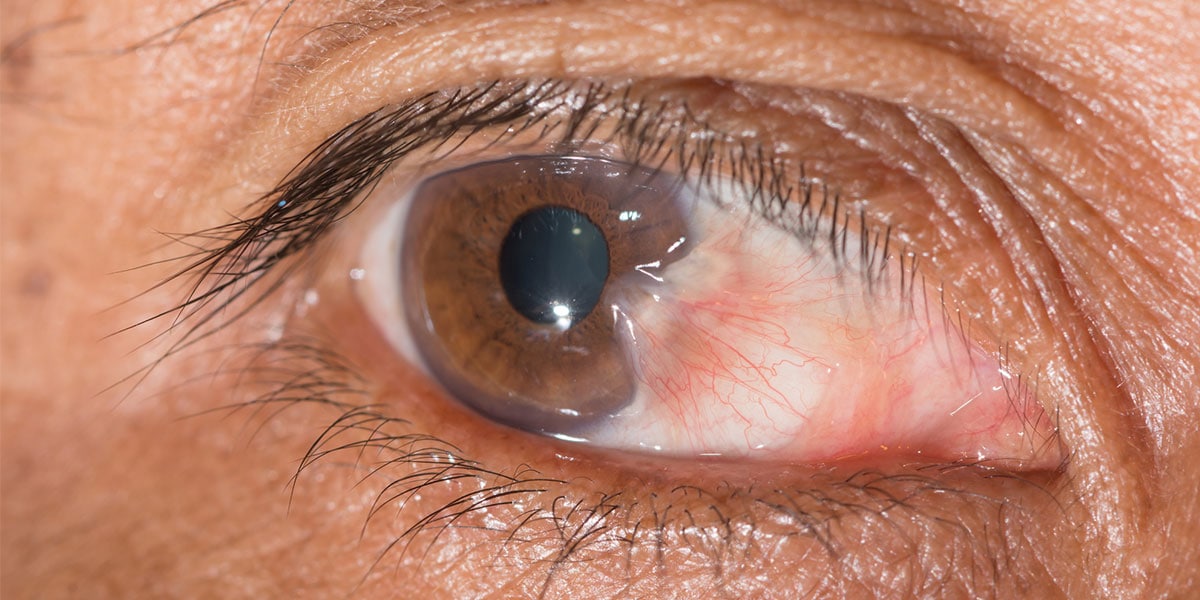
You're in the very best of hands with our world-class LASIK, cataract, & retinal surgeons who are dedicated to providing compassionate care, and pioneering advanced in ophthalmology, including these conditions:
Meet the Assil Gaur Eye Institute Doctors
We are whole-heartedly committed to delivering the most exceptional care anywhere, and providing state-of-the-art treatments for all eye conditions including glaucoma, macular disease, dry eye, and diabetic eye conditions, just to name a few.
Dr. Kerry Assil
Medical Director, Refractive & Cataract SurgeonDr. Avneet K. Sodhi Gaur
Director of Glaucoma Services, Refractive & Cataract Surgeon, AestheticsDr. Svetlana Pilyugina
Director of Retinal Division, Vitreo-Retinal SurgeonDr. Adeleh Yarmohammadi
Director of Oculoplastics, Anterior Segment & Glaucoma SurgeonDr. Rafi Israel
Oculoplastics SurgeonDr. John May
Comprehensive Eye Care, Pre/Post Operative Surgical CareDr. Lindsay Harris
Comprehensive Eye Care, Pre/Post Operative Surgical CareDr. Sahar Zokaeim
Doctor of OptometryDr. Natalie Sarukhanian
Doctor of Optometry
-

LeBron James
Los Angeles LakersLeBron James is one of the NBA's greatest of all time. He absolutely depends on his vision to perform at the very highest level. That's why he's an AGEI LASIK surgery patient.
-
LeBron James
Los Angeles LakersLeBron James is one of the NBA's greatest of all time. He absolutely depends on his vision to perform at the very highest level. That's why he's an AGEI LASIK surgery patient.
-
Anthony Davis
Los Angeles Lakers -
Chris Paul
Phoenix Suns -
Paul George
Los Angeles Clippers -
Marlee Matlin
Actress -
Michelle Williams
Actress -
Dwyane Wade
Miami Heat (retired) -
LaToya Jackson
Entertainer -
Lorenzo Lamas
Actor -
Philip Bailey
Earth Wind and Fire -
Gary Sinise
Actor -
Rip Hamilton
Former NBA player -
John Salley
Commentator, former NBA player -
Blair Underwood
Actor -
Tom ArnoldComedian/Actor
-
Troy EvansActor
-
John NobleActor
-
Isaac EddyActor/Director/Teacher
-
Barbara RushActress
-
John EmersonUS Ambassador to Germany
-
Shane MosleyFormer professional boxer
-
Cuttino MobleyFormer NBA player
-
Seth GordonDirector
-
Maurice EvansFormer NBA player
-
Wang LuoyongActor
-
Jesse MetcalfeActor
-
Ananda LewisTelevision Personality
-
Rob BrydonComedian
-
Clifton Collins Jr.Actor
-
Malinda WilliamsActress
-
Adrienne FrantzActress
-
Brad PittActor
-
LeBron JamesLos Angeles Lakers
-
Anthony DavisLos Angeles Lakers
-
Chris PaulPhoenix Suns
-
Courtney CoxActress
-
Paul GeorgeLos Angeles Clippers
-
Marlee MatlinActress
-
Michelle WilliamsActress
-
Dwyane WadeMiami Heat (retired)
-
Jalen BrunsonDallas Mavericks
-
Mychal ThompsonCommentator, former NBA player
-
LaToya JacksonEntertainer
-
Lorenzo LamasActor
-
Philip BaileyEarth Wind and Fire
-
Gary SiniseActor
-
Rip HamiltonFormer NBA player
-
John SalleyCommentator, former NBA player
-
Blair UnderwoodActor
-
Tom ArnoldComedian/Actor
-
Troy EvansActor
-
John NobleActor
-
Isaac EddyActor/Director/Teacher
-
Barbara RushActress
-
Rudy GarciduenasBlue Man Group
-
John EmersonUS Ambassador to Germany
-
Shane MosleyFormer professional boxer
-
Cuttino MobleyFormer NBA player
-
Seth GordonDirector
-
Maurice EvansFormer NBA player
-
Cheri BlauwetPhysician and Wheelchair Racer
-
Wang LuoyongActor
-
Jesse MetcalfeActor
-
Jeana WilsonActress
-
Courtnee DraperActress
-
Ananda LewisTelevision Personality
-
Robyn JohnsonProducer
-
Rob BrydonComedian
-
Cary SchumanActor
-
Roland Buddy Lewis Jr.Comedian
-
Clifton Collins Jr.Actor
-
Malinda WilliamsActress
-
Chad JeffersMusician
-
Adrienne FrantzActress
-
Franco CarlottoBody Builder
-
Brad MatesMusician
-
David PichetteMusician
-
Mike MelaconProfessional Baseball Player
-
Nautica de la CruzRadio Personality
-
Lynn ConkwrightBody Builder
-
Nely GalanProducer
-
Roxanne GallaActress